OX-A
Clear anodizing
OX-A is a clear aluminium anodizing treatment in compliance with MIL-A-8625 Type II, and ISO 7599.
OX-A anodizing treatment is composed of an electrolytic aluminium oxidation process. It is performed by dipping the substrate in a sulphuric acid solution at 20°C and by applying direct electric current.
During the process, the surface of the aluminium part is transformed, creating a protective film of aluminium oxide with a typical thickness of 10-20µm.
The OX-A treatment protects treated parts against corrosion and wear. The corrosion resistance of parts anodized with OX-A passes 336 hours in salt spray in compliance with MIL-A-8625.
The treatment is used to protect automatic machinery components, in the medical field, home appliances, and industrial components.

FEATURES
CORROSION RESISTANCE
The OX-A film protects the base material from corrosion, exceeding 336 hours of salt spray exposure in compliance with the requirements of MIL-A-8625.
RESISTANCE TO LIGHT WEAR AND SCRATCHES
The layer of aluminium oxide formed by the OX-A treatment makes it possible to achieve high scratch resistance and resistance to minor wear phenomena.
ECONOMICAL
Compared to other aluminium anodizing treatments, OX-A is more economical thanks to the high efficiency process.
LOW-FRICTION VARIANT OX-A-PTFE
To lower the coefficient of friction and provide anti-adhesion, the OX-A treatment can be impregnated with PTFE nanoparticles.
AVAILABLE COLORS
OX-AN - Black Color
OX-A treatment can be colored in deep black. The deep black allows the color to be equalized on different aluminium alloys.

OX-AB - Blue Color
OX-A treatment can be colored blue. The color shade depends on the treated aluminium alloy. The picture is an indicative representation of the color obtainable on alloy 7075 and alloy 6061.

OX-AR - Red Color
The OX-A treatment can be colored red. The color shade depends on the treated aluminium alloy. The picture is an indicative representation of the color obtainable on alloy 7075 and alloy 6061.

TECHNICAL SPECIFICATIONS
Composition and applicable standards
| Composition | |||
|---|---|---|---|
| The OX-A treatment transforms the aluminium substrate into a compact layer of aluminium oxide. The coating composition is thus strongly dependent on the starting alloy. | |||
| Al | O | S | Impurities |
| 20÷40% | 50÷70% | 3÷5% | Depending on the alloy |
| Technical standards |
|---|
| ISO 7599 |
| MIL-A-8625 | Type II |
| RoHS compliance |
|---|
| RoHS compliant. No restricted substances present in amounts greater than the maximum tolerated concentrations. |
| REACH compliance |
|---|
| REACH compliant. No SVHCs present in amounts higher than 0.1% by weight. |
Anodizable alloys
| Wrought alloys | Characteristics | |
|---|---|---|
| Containing high percentages of copper or zinc | Corrosion resistance | ★★★☆☆ |
| Maximum thickness | ★★★★☆ | |
| Other alloys | Corrosion resistance | ★★★★★ |
| Maximum thickness | ★★★★★ | |
| Casting alloys | Characteristics | |
|---|---|---|
| Alloys with Si>8% or Cu>2% | Corrosion resistance | ★☆☆☆☆ |
| Maximum thickness | ★☆☆☆☆ | |
| Die castings with Si<8% or Cu< 2% | Corrosion resistance | ★★☆☆☆ |
| Maximum thickness | ★☆☆☆☆ | |
| Other alloys | Corrosion resistance | ★★★☆☆ |
| Maximum thickness | ★★★☆☆ | |
Treatment thickness and aesthetic appearance
| Coating thickness | |
|---|---|
| Standard thickness | Tolerance |
| 15µm | ±5µm |
30% of the treatment thickness is due to external film growth and 70% is due to film growth in the surface of the aluminium part. The radial dimensional increase can thus be calculated as 30% of the treatment thickness.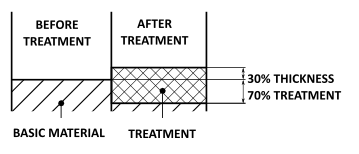 | |
| Uniform thickness over the entire external surface. Reduced thickness in holes. | |
| Aesthetic appearance |
|---|
| Semi-polished appearance with light grey color. The color depends on the base alloy. Reproduces the morphology of the machined part. |
| Option of black coloration in the OX-AN version |
| Possibility of blue coloring in the version OX-AB |
| Possibility of red coloring in version OX-AR |
Tribological properties
| Wear resistance |
|---|
| Resistance to moderate wear and scratches. To meet higher demands, OX-HS and OX-W treatments can deliver very high wear resistance. |
| Coefficient of friction |
| The OX-A-PTFE variant consists of an impregnation treatment of the anodizing layer with nanometric PTFE particles. This impregnation provides a non-stick, self-lubricating surface with a low coefficient of friction.. |
Chemical properties
| Corrosion resistance | ||
|---|---|---|
| The OX-A treatment makes it possible to achieve high corrosion and oxidation resistance. Exceeds requirements for resistance to the accelerated salt spray corrosion test in compliance with MIL-A-8625F Type II. | ||
| Corrosion resistance value | Substrate material | |
| NSS to MIL-A-8625F 3.7.1.2 | ||
| ≥336 hours | 6000 alloy | |
| Chemical resistance | |
|---|---|
| Chemical compatibility values. The actual environmental resistance must anyway be tested in the field. | |
| Hydrocarbons (e.g. petrol, diesel, mineral oil, toluene) | |
| Alcohol, ketones (e.g. ethanol, methanol, acetone) | |
| Neutral saline solutions (e.g. sodium chloride, magnesium chloride, seawater) | |
| Dilute reducing acids (e.g. citric acid, oxalic acid) | |
| Acid oxidizing agents (e.g. nitric acid) | |
| Concentrated acids (e.g. sulphuric acid, hydrochloric acid) | |
| Dilute bases (e.g. dilute sodium hydroxide) | |
| Base oxidizing agents (e.g. sodium hypochlorite) | |
| Concentrated bases (e.g. concentrated sodium hydroxide) | |

 Technical datasheet
Technical datasheet